Since the discovery of liposomes in the 1960s, lipid nanoparticles (LNPs) have become a mainstream platform for drug delivery systems. Leveraging nanotechnology, LNPs enable the stable and efficient delivery of a wide range of therapeutic drugs. In recent years, LNPs have gained global attention as a critical component of COVID‑19 mRNA vaccines, where they play an essential role in effectively protecting mRNA and delivering it into cells.
Liposomes represent the first generation of LNPs and the earliest nanoscale drug delivery platform, successfully advancing from conceptual development to clinical application. Since their discovery, substantial technological progress has been made, ranging from liposome formulation to targeted liposome design, to enhance their functionality. Numerous liposome‑based drugs have been approved, including Doxil (doxorubicin), an anticancer agent for ovarian cancer, and Epaxal, a protein‑based vaccine against hepatitis.
Early liposome manufacturing methods were relatively complex, requiring organic solvents, and typically suffered from low drug encapsulation efficiency and poor scalability. In addition, conventional liposomes faced challenges such as short circulation time in the bloodstream, insufficient stability, and lack of selective targeting.
To address these limitations, the modification and engineering of liposomal particles have driven the development of more advanced lipid nanoparticles. Key principles underlying advanced LNP design include structural and application‑related aspects. Structural factors comprise biocompatibility, fluidity, phase behavior and transition temperature, surface charge (zeta potential), particle size, encapsulation efficiency, and stability. Application‑related factors include toxicity, circulation time, cellular uptake, and cargo release.
To date, LNPs have achieved remarkable improvements in efficacy, selectivity, and in vivo drug distribution, while reducing the toxicity and limitations associated with conventional drug delivery systems.
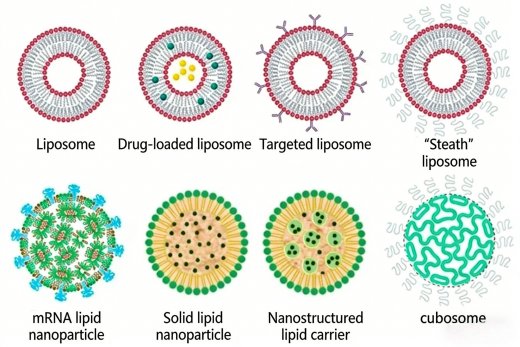
Different types of LNPS have their own advantages and disadvantages, which determine their application in various therapeutic drugs. A new type of drug is nucleic acid therapy drugs, which show potential in treating a variety of diseases. The first approved drug of this type is Patisiran (ONPATTRO), a small interfering RNA (siRNA) using LNP preparations, which is used to reduce the formation of transthyretin protein in the liver for the treatment of hereditary transthyretin-mediated amyloidosis.
Perhaps the most well-known latest application of LNP is as a delivery vector in the approved COVID-19 messenger RNA (mRNA) vaccines developed by Pfizer/BioNTech and Moderna. These vaccines deliver mRNA encoding the SARS-CoV-2 spike protein to the cytoplasm of the host cell, where it is translated and acted as an antigen to induce an immune response against the virus.
LNP formulations for mRNA typically consist of multiple components. The primary functional component is ionizable cationic lipid, which usually accounts for approximately 50% of the entire structure. It facilitates nucleic acid encapsulation during formation, helps maintain a neutral charge in circulation, and promotes intracellular delivery. Cholesterol constitutes about 40% of the structure. Owing to its hydrophobicity, it contributes to rigidity, formulation stability, and supports controlled release. Next is the PEG-lipid component, at around 1–2%, which helps control and maintain particle size and prolongs circulation time by preventing opsonization in the blood.The remaining approximately 10% of the formulation consists of structural lipid components such as DSPC and DOPE, which contribute to the overall structural stability of the formulation as well as stability during manufacturing and long-term storage. Optimization of LNPs can significantly improve the safety and efficacy of LNP-mRNA-based therapeutics. This includes innovations in ionizable and biodegradable lipids for different application routes; optimization of LNP-mRNA composition, such as varying lipid ratios or lipid-to-mRNA ratios; novel stealth lipids; and improved off-target effects through tissue-specific localization and active targeting of LNP-mRNA therapeutics to specific cell types and organs, paving the way for new applications in hard-to-target tissues.
LNP-mRNA formulations are commonly produced using staggered Y-junction microfluidic devices. One inlet stream contains mRNA in an acidic buffer, and the other inlet stream consists of lipids dissolved in ethanol. The two streams meet and mix in the mixing channel and exit the device as a single stream. Downstream of the microfluidic device, mRNA strands become encapsulated within LNPs. At this stage, tangential flow filtration (TFF) is used to remove all components outside the mRNA-LNPs, including solvents used in the microfluidic step, and to perform buffer exchange.
Tangential flow filtration (TFF) is a common unit operation in biopharmaceutical production, used for the purification, concentration, and buffer exchange of target products. Compared with dead-end filtration, TFF reduces the accumulation of contaminants on the membrane surface and the formation of a gel layer by sweeping generated through the feed flow tangentially and parallel to the membrane surface, thereby alleviating membrane fouling and improving the processing capacity per unit membrane surface area. Moreover, owing to its continuous recirculating flow mode, the product in the permeate or retentate can be recovered as the target product.
Based on membrane structure design, common TFF formats include flat-sheet membrane cassettes and hollow fiber membranes. In addition, factors affecting the performance of TFF steps include configurations such as membrane material, membrane pore size/molecular weight cutoff (MWCO), pump drive type, and flow path design, as well as process parameters such as transmembrane pressure (TMP), shear force, and filtration flux. These need to be selected and optimized according to the characteristics of the target product, especially for stress-sensitive products such as mRNA-LNP during manufacturing processes.
Hollow Fiber vs. Membrane cassette
Hollow fiber membranes are tubular fiber membranes with filtration pores distributed on the fiber wall. A certain number of fibers are potted inside a tubular housing to form a filtration module. The lumen of each hollow fiber is an open flow channel, so the fluid flow inside hollow fibers is close to a gentle laminar flow state.
Flat-sheet membrane cassettes consist of multiple layers of flat membranes stacked together and separated by spacer screens to form flow channels. The fluid flow pattern inside flat-sheet cassettes is closer to turbulent flow, which enhances the scouring effect on the membrane surface.
In most cases, for shear-sensitive target products, using hollow fiber-based tangential flow filtration is more conducive to protecting the structural integrity of the product.In contrast, the turbulent flow promotion and higher operating pressure tolerance of flat-sheet membrane cassettes may lead to higher flux under otherwise similar conditions.
For mRNA-LNP, hollow fiber filters are preferentially recommended for evaluation to ensure the yield of intact products, and good performance can be achieved for most product types.For more structurally stable products, flat-sheet membrane cassettes may deliver higher flux without compromising product integrity.

Pump selection
Pumps serve as the source for generating "tangential flow" and the transmembrane pressure required to drive the filtrate through the membrane. Currently, various types of pumps are used in tangential flow filtration, including peristaltic pumps, diaphragm pumps, and magnetic drive centrifugal pumps. Peristaltic pumps drive fluid flow by continuously compressing a flexible tube. Since the fluid does not come into contact with the pump itself, the risk of contamination is minimal.
Diaphragm pumps operate on a principle similar to the beating of a heart, utilizing four-piston diaphragm technology. They pump the fluid gently through a slow "heartbeat" motion. Each stroke of the four diaphragms is generated by an eccentric shaft connected to a motor. This operating mode allows the pump to transfer the fluid gently, safely, and smoothly. Magnetic drive centrifugal pumps are based on magnetic levitation technology. The pump impeller is suspended without contact inside a sealed housing and is driven by a magnetic field from the motor, generating a continuous and stable flow. Depending on the specific characteristics of the target product and the processing scale, the appropriate pump type and size can be selected based on certain parameters, such as specific output flow requirements and the choice between single-use or reusable options. For example, when dealing with very low sample volumes, a peristaltic pump may be chosen to minimize the system's minimum operating volume requirements. If the target product is unstable and susceptible to damage even from the gentle compression of a peristaltic pump, a diaphragm pump or a magnetic drive centrifugal pump could be considered.
Technological parameter
The main process parameters in tangential flow filtration include tangential flow velocity (also known as crossflow velocity), transmembrane pressure (TMP), filtrate flux, and shear stress, all of which interact with one another.
Tangential flow velocity refers to the actual recirculation velocity across the membrane surface and serves as the source of sweeping action on the membrane. The higher the tangential flow velocity, the stronger the sweeping effect, which reduces the likelihood of membrane fouling. However, tangential flow velocity is also positively correlated with the shear stress generated within the flow channel. Therefore, the selection of tangential flow velocity should ensure that the corresponding shear stress does not compromise product quality. As a rule of thumb, a shear rate of approximately 3000–4000 s⁻¹ can be used as an initial experimental reference and adjusted based on filtrate flux and product quality results.
On the other hand, TMP is the direct driving force for filtrate formation. It depends on factors such as the inlet pressure and the pressure drop across the filter. Within a certain operating range, TMP is positively correlated with filtrate flux. However, higher TMP conditions also make membrane fouling more likely. In ultrafiltration processes, constant TMP operation is sometimes chosen, but as concentration proceeds, constant pressure conditions may not yield a stable filtrate flux. Therefore, constant flow—or constant filtrate flux—can be a viable alternative.
During process development, the recording and analysis of all process parameters is an important feature of advanced tangential flow filtration systems. This not only aids in troubleshooting to achieve better and more reproducible process performance but also serves as the foundation for subsequent process scale-up and regulatory audits.
The tangential flow filtration is a fundamental unit operation in the mRNA-LNP process flow. Selecting the appropriate tangential flow filtration form, system configuration, and process parameters, which are the key influencing factors of the process, is crucial for achieving process success. Repliculin provides tangential flow filtration solutions based on different filter forms (hollow fibers and flat membrane packages), different pump forms (peristaltic pumps, diaphragm pumps, magnetic centrifugal pumps), and different usage strategies (one-time use and reuse). It has complete data records and auxiliary analysis functions for a series of systems ranging from scale-down process development to scale-up clinical trials and commercial production, and can be configured according to GMP requirements of the production environment, thus meeting the requirements of different development stages of mRNA-LNP.





